Polyethylene glycol (PEG), a compound composed of repeated ethylene glycol units [-(CH2CH2O)n], is also known as Macrogols. PEG and PEG derivatives have the characteristics of non-irritation, good water solubility, good compatibility, etc. . At the same time, they possess excellent lubricity, moisture retention and dispersibility so that they can be used as adhesives, antistatic agents and softeners, and widely applied in cosmetics, pharmaceuticals, chemical fibers, rubber, plastics, paper, paint, electroplating, pesticides, metal processing and food processing industries.
Synthesis of PEG
PEG can be synthesized via anionic polymerization of ethylene oxide and any hydroxyl initiators. Hydroxyl group could be from water or ethylene glycol or any diols. They can also be derived from epoxyethane by ring-opening polymerization. Usually, the polymerization process produces a family of PEG molecules with wide Gaussian’s distribution of molecular weights. PEG bears only two functional groups limiting the scope for further derivatization with other residues or targeting ligands. Therefore, commercial PEGs are available with different degrees of polymerization and activated functional groups.
PEG Properties
PEG earns its fame because of its high structure flexibility, biocompatibility, amphiphilicity, devoid of any steric hindrances, and high hydration capacity. PEG is soluble in water, ethanol and many other organic solvents, like DMF, dichloromethane, toluene, acetonitrile, acrylonitrile, etc. . It shows little toxicity and can be eliminated from the body intact by either the kidneys (for PEGs 20 kDa). Other than that, PEG is also non-irritating and stable to heat, acid and alkali, thus it can be widely used in various pharmaceutical applications.
PEG series products have different properties depending on the different molecular weights (Mw). From the perspective of appearance, PEG with Mw of 200-700 are normally liquid at room temperature, and those with Mw > 700 will gradually change from semi-solid to soft solids (Mw between 1000~2000), and finally to hard crystalline solids (Mw > 2000). On the other hand, as the Mw increases, the water solubility, vapor pressure, water absorption, and solubility in organic solvents of PEG will correspondingly decrease, while the freezing point, relative density, flash point and viscosity will increase accordingly. The following table briefly lists several properties of PEG products with different Mw.
PEG and PEG Derivatives
PEG-NHS ester; PEG-aldehyde; PEG-maleimide; PEG-hydrazide; PEG-amine; PEG-alkyne; PEG-azide; PEG-vinyl sulfone; PEG-thiol.
PEG, a polyether backbone polymer, is synthesized by ring opening polymerization of ethylene oxide using in common cases or usually methanol or water as an initiator. The reaction gives products with one or two end chain hydroxyl groups termed monomethoxy-PEG (mPEG-OH) or diol-PEG (HO-PEG-OH), respectively (Fig. 2).
The lone hydroxyl group in the case of the methoxy form, or the two in the case of PEG diol, can be modified to be reactive towards different chemical groups by several activation strategies. Nowadays, a number of activated PEGs are commercially available. These derivatives are commonly monofunctional, either in their linear or branched structure. The chemical structures of several key PEG derivatives are shown in Figure 2. These derivatives are mainly known by their Mw.
PEGylation Technique
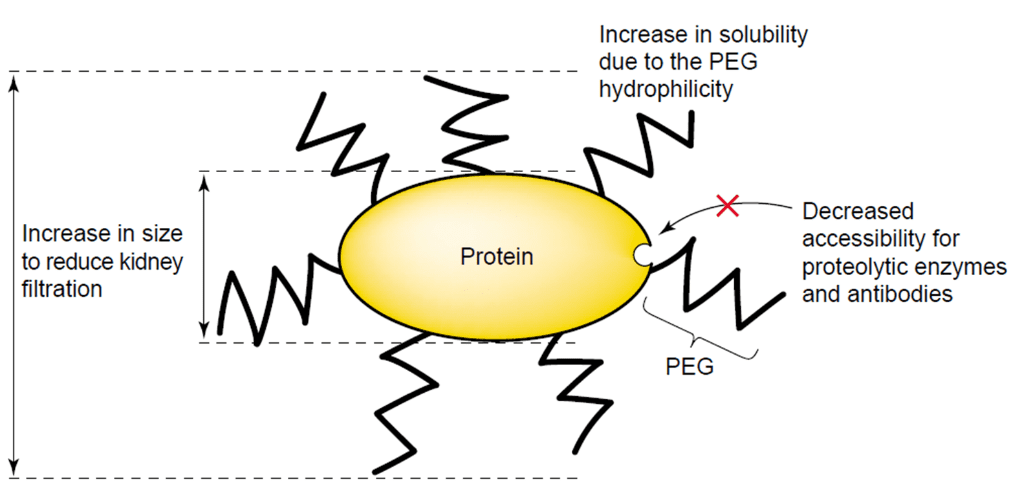
PEGylation is defined as the covalent attachment of poly(ethylene glycol) (PEG) chains to bioactive substances, such as nanoparticles, proteins, peptides or non-peptide molecules. This technology emerged gradually with progress in field of biologicals and polymer chemistry and was exploited to improve pharmaceutical applications of a wide range of therapeutics. Nowadays, PEGylation has become the leading approach for overcoming most of the limits of biologics and numbers of FDA approved PEGylated products like enzymes (bovine adenosine deaminase and urate oxidase), cytokines (interferon-α2a, interferon-α2b), granulocyte colony stimulating factors, hormones (epoetin-β), antibodies and their fragments and other organic molecules (pegvisomant, pegatinib) are on the market.
References
1.D’souza, A. A.; Shegokar, R., Polyethylene glycol (PEG): a versatile polymer for pharmaceutical applications. Expert Opinion on Drug Delivery 2016, 13 (9), 1257-1275.
2.Veronese, F. M.; Pasut, G., PEGylation, successful approach to drug delivery. Drug discovery today 2005, 10 (21), 1451-1458.
3.Fruijtier-Pölloth, C., Safety assessment on polyethylene glycols (PEGs) and their derivatives as used in cosmetic products. Toxicology 2005, 214 (1-2), 1-38.